|
His idea was not taken seriously at the time, however, and it was many centuries before the concept of the atom would be revived. He used the term atomos, Greek for “indivisible,” from which the modern term “atom” is derived. The first person to propose that everything on Earth is made up of tiny particles was the Greek philosopher Democritus, around 450 B.C. Together, these three particles account for the mass and the charge of an atom. Each atom is made up of three subatomic particles: protons, neutrons, and electrons. In this case, they can lose electrons and become positively charged ions called cations or gain electrons and become more negative anions.Īll matter is composed of atoms, the smallest individual units of elements. However, depending on the electron configuration, they can be unstable as single atoms. Generally, an atom contains an equal number of protons and electrons, which cancels out the positive and negative charges, leading to no net charge. Electrons remain near the nucleus because they are attracted to the positive charge of protons. This cloud is mostly empty space and makes up the vast majority of an atom's volume. While both contribute to the mass, carrying one atomic mass unit each, it's the number of protons that determines the specific element, referred to as the atomic number.Įlectrons, a third type of subatomic particle, are negatively charged and move rapidly in a cloud around the nucleus. Protons are positively charged while neutrons are uncharged, neutral. Two of these particle types are clustered in a central nucleus. 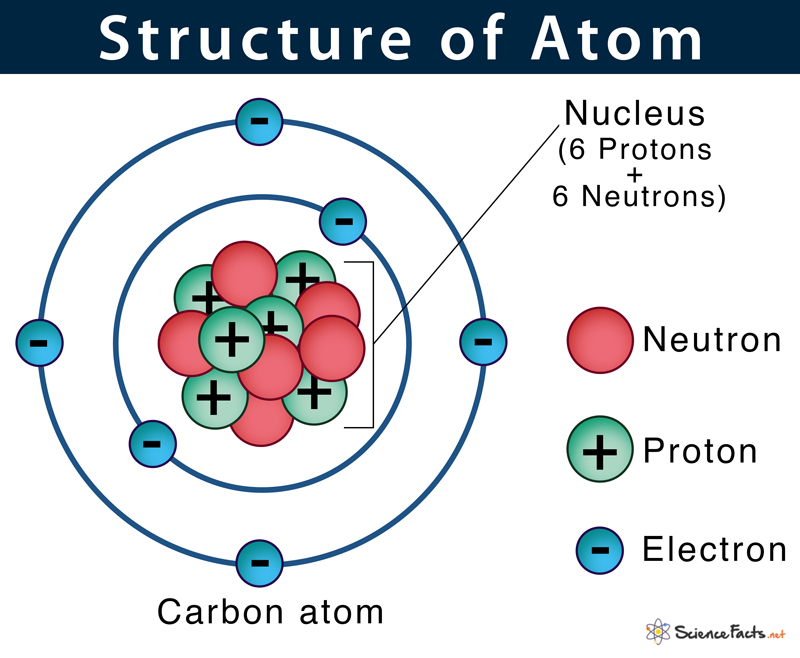
These differences are found in the subatomic particles that make up each atom. Each element, like potassium, consists of a unique type of atom that differs from the atoms of other elements. 
Elements are substances with unique physical and chemical properties that cannot be broken down into other substances by chemical reactions.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
